Tour of the Clinical Study Tracker
Part 1: How can I use rapid prototyping to help me decide whether CST is for me?
The rapid prototyping phase allows potential users to test out their own clinical study activities and process steps in a demo version of the Clinical Study Tracker without committing it to an actual database. They will be able to view what their application and forms might look like in a matter of minutes. This is simply done by a study coordinator sitting with their IT administrator or someone who has enough technical skill to alter the example XML configuration files that come with the release. During the rapid prototyping phase, a study coordinator should answer these questions:- What word do we want to use to describe what we're studying? "case study" "volunteer"? "participants?"
- What kinds of demographics about subjects do we want to maintain? The clinic they're registered? The county they live in? Their ethnicity? Age? Gender?
- What medical tests or activities do we need to follow for each of these participants?
- What process steps might each one of these tests or activities have to go through for this study to complete the process?
- What process steps do we perform for this activity?
- Do we expect the steps for this activity to always be in chronological order?
- Is one person going to log this data or will it be shared by multiple people that I wish to have as designated users?
- Do you want to allow users to fill in a field but allow previous fields to be blank?
In a short time period, both the study coordinator and the IT administrator can decide whether the Clinical Study Tracker is the ideal and cost saving tool you have been searching for. In addition, clinical studies may either choose to only use CST to rapidly prototype an application or in-house developers can then elect to create their own application from scratch using the prototype as a guide.
Figure Prototype-1 shows how a clinical study would begin using the tool.
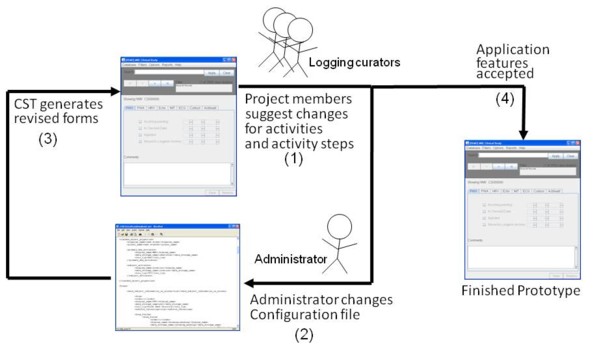
Fig. Prototype-1: Rapid prototyping with CST. Study coordinators suggest activities (1) to administrators, who make appropriate changes to a configuration file(2). The administrators use CST to generate demonstration versions of the logging tool (3). The study coordinators try out the generated application and provide feedback to the administrators. When the users are satisfied with the application features, prototyping ends (4).
Part 2: Yes, CST for me! Using it in production
Administrators can use the configuration file they made in the prototyping phase to generate production versions of the tools. Figure Production-1 shows how users would use CST.

Fig. Production-1: Using CST in production. Using CST in production. After administrators install MySQL Server 5.0 or higher, they can run the administration tool with the configuration file produced in the prototyping phase (1). The administrators import subject demographic data and user identities(2). Users can then begin manually creating the forms (3) or importing log data from spreadsheets (4). Using the curated data, they can produce progress reports as spreadsheets (5) or graphs (6). They can product audit trails of changes (7) or export activity data to analyse in other applications such as Excel (8).
First, they must ensure that they have installed MySQL server 5.0 or higher on some target user system. Then they can run the administration tool and use its "Create Database" command to create an empty MySQL database.
Administrators will use the administration tool to do two other tasks:
- load demographic data about all the subjects from a tab-indented text file.
- define login names and passwords for people who will be using the Logging Tool.
Users can produce a spreadsheet containing an identifier field for the subject (eg: an NHS number) and a column for each of the demographic fields defined in the configuration file (eg: "Location", "Gender"). Administrators can export the spreadsheet to a tab-indented text file and use the Administration tool to import data for the subjects.
Next, they can define user identifies for team members who will be logging data (eg: "kgarwood", "jsmith", etc). Users will then be ready to use the Logging Tool. They can use the data entry forms in the application to manually update the progress of a case study through an activity. They can also import logged dates from tab-indented spreadsheets. Project members can use the report features of the Logging Tool to produce progress charts.
Occasionally users may want to add additional activities. Administrators can support new activities by following two steps. First, they can add new definitions of activities and activity steps to the Configuration File. Then they can use the "Synchronise" feature of the Administration Tool to ensure that the database maintains corresponding tables and fields to hold new kinds of record data.
Throughout the course of a clinical study, project members may use CST to track the progress of case study members through multiple activities. They may acquire update progress data for each participant either by using the forms or importing log data from spreadsheets. Each import spreadsheet holds data for a single activity. Periodically the curators may produce progress reports to inform other project members about how many participants have been processed for given activity steps. The application produces graphs and spreadsheets of progress data based on filters based on some demographic aspect such as location, based on dates for activity steps or a combination of both.
CST keeps track of what changes each logging curator makes to an activity record. The audit trail reports can show all changes made to a given subject or all changes made by the current user. All audit trails appear in reverse chronological order. In some cases, users may want to export activity data to analyse in other applications. For example, users may want to create sophisticated graphs based on activity data. They can use CST to export data for an activity to a tab-delimited text file that can be imported into Microsoft Excel.
Part 3: Enhancing the Software
Eventually your project may want to add special features to CST. The software allows developers to create custom plugins which can be accessed through the menu of the logging tool. You may even want to start CST from some other application which manages clinical data. Figure Enhance-Software-1 shows how the software can be used in new ways by your developers.
Fig. Enhance-Software-1: Your developers can add plugins to the Logging Tool. They can also start the Logging Tool from another application. CST allows your project to use multiple Logging Tools to log data for different groups of activities.
If you have developers on the team, let them read the section about creating plugins.
Part 4: Retiring the application and migrating data
There are at least two reasons why projects may want to stop using CST:- the study is finished and user wants to store data in long-term data archives
- their clinical study expanded or evolved and can no longer be supported by the software.
Figure Data Migration-1 shows how CST supports a simple data migration procedure.
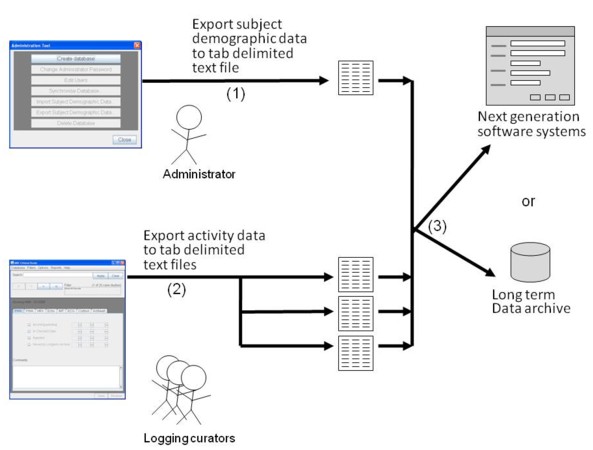 Figure Data Migration-1: Project stops using CST. Administrators can
use the administration tool to export all the subject demographic data to a
tab-delimited file (1). Users can export all the progress data for each activity
to a corresponding tab-delimited text file (2). The text files can be moved to
some simple long term storage facility or they can be used to transfer data to
another application.
Figure Data Migration-1: Project stops using CST. Administrators can
use the administration tool to export all the subject demographic data to a
tab-delimited file (1). Users can export all the progress data for each activity
to a corresponding tab-delimited text file (2). The text files can be moved to
some simple long term storage facility or they can be used to transfer data to
another application.
Author: Sheila Raynor
(c)2010 Medical Research Council. Licensed under Apache 2.0.
